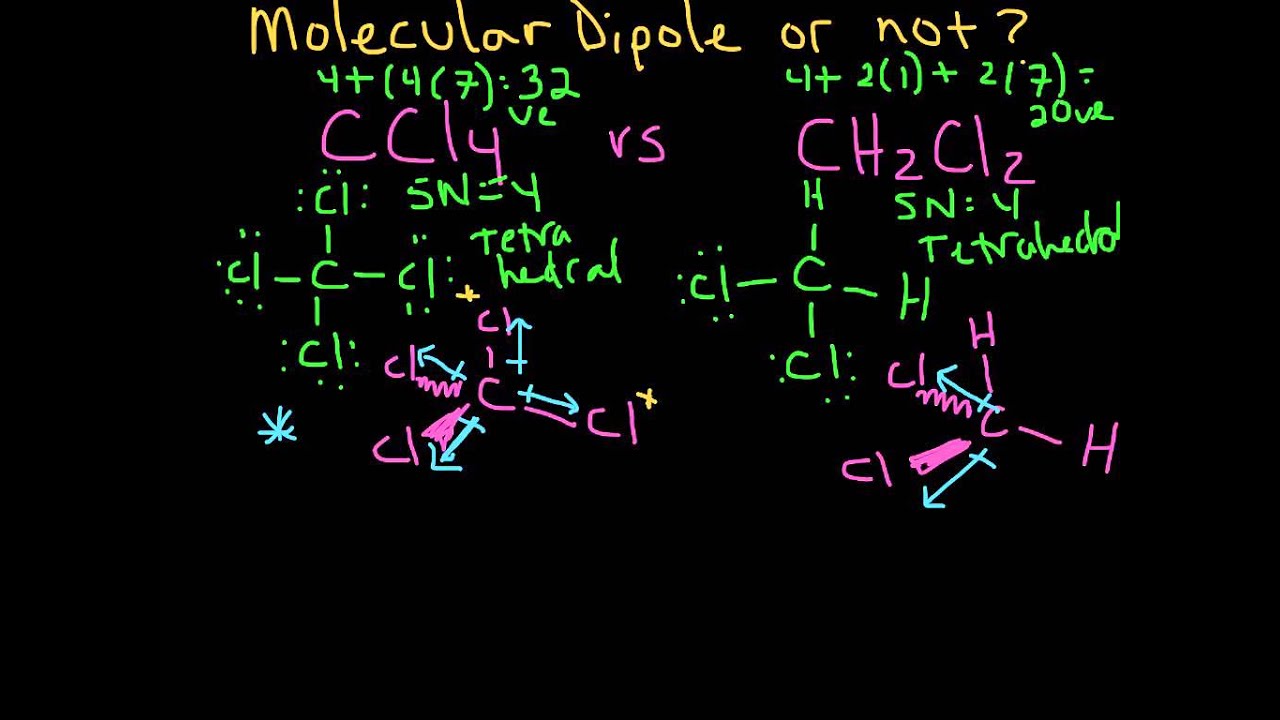halides - Why does chloromethane have a larger dipole moment than chloroform? - Chemistry Stack Exchange

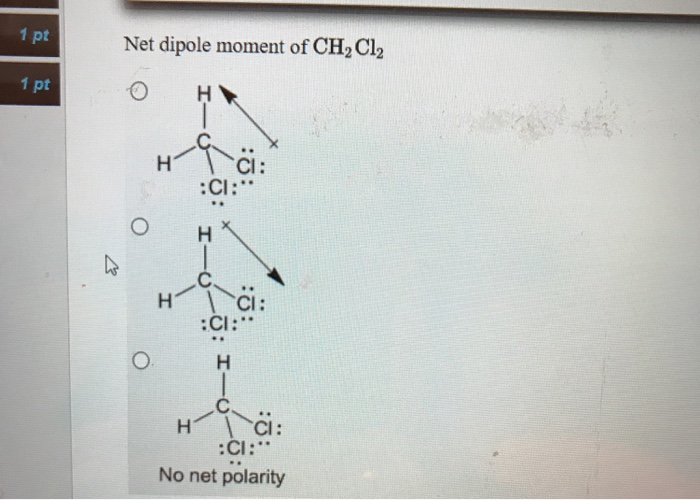
Which of the following show the correct direction of the dipole moment for CH2Cl2? | Homework.Study.com

Which of the following show the correct direction of the dipole moment for CH2Cl2? | Homework.Study.com

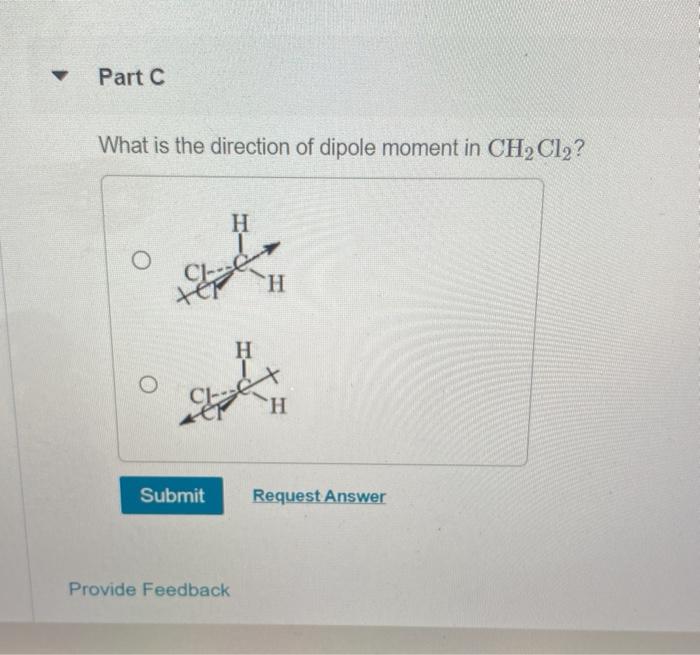
Outline a three-dimensional representation of the given molecule and indicate the direction of any net dipole for the given molecule. CH2Cl2 | Homework.Study.com

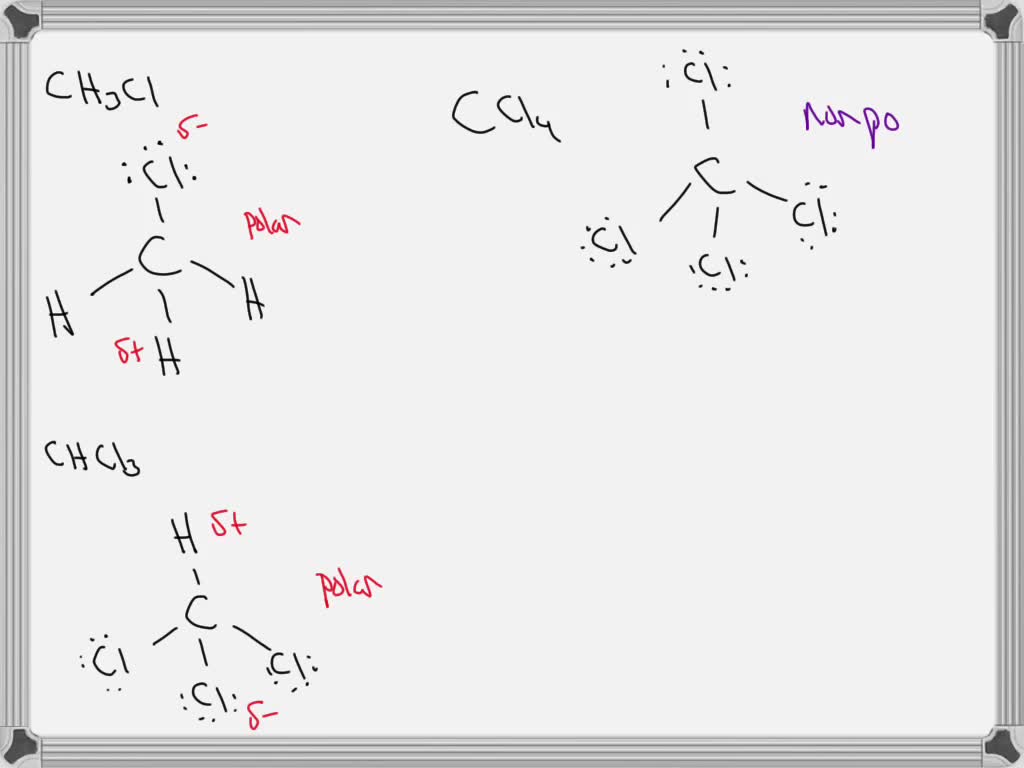
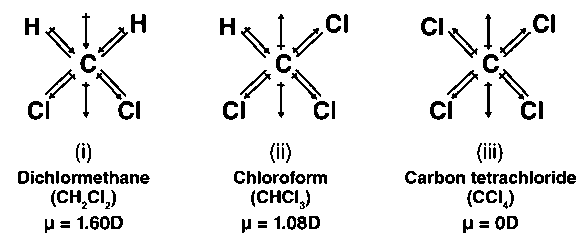
how to calculate dipole moment of CH2CL2 ,CHCL3,CCL4 - Chemistry - Haloalkanes and Haloarenes - 6575366 | Meritnation.com



![Best Explanation: CH2Cl2 polar or nonpolar [N0#1] - Science Education and Tutorials Best Explanation: CH2Cl2 polar or nonpolar [N0#1] - Science Education and Tutorials](http://sciedutut.com/wp-content/uploads/2021/05/IS-CH2Cl2-polar-or-nonpolar-1.png)